Shalini Gupta
| PhD, Chemical Engineering, 2007 North Carolina State University M.S., Chemical Engineering, 2004 North Carolina State University B.Tech., Chemical Engineering, 2002 Institute of Technology, Banaras Hindu University, India Present Site |  |
Research Focus: Biomaterials and Biosensors Based on Live Cell-Colloidal Particle Interactions
The overall focus of my research is to develop new types of immuno- and cell biosensors by studying molecular, particle-particle and cell-particle interactions at the colloidal scale (1 nm – 10 µm). The overarching goal of my first research project is the controlled fabrication of bio-inorganic composite materials made from live cells and functionalized nanoparticles using engineered principles of dielectrophoresis. Dielectrophoresis is a phenomenon wherein a force is exerted on a dielectric (charged or uncharged) particle in a non-uniform electric field. An AC electric field is applied across a mixture of cells (yeast and mouse fibroblast) and lectin-functionalized magnetic particles to obtain permanent assemblies, viz. 1-D chains and 2-D membranes (as shown in Fig. 1). We can assemble structures upto 2 x 2 cm2 from micron sized cells in a single step. These structures can be easily manipulated using external magnetic fields. These types of material have potential applications in sensors, tissue engineering, bioreactors, and etc.

Figure 1: Permanent 1-D chains and 2-D membrane fabricated from live yeast cells.
My second project involves the development of inexpensive silver enhanced nanoparticle-based immunoassays and their optimization using a fundamental adsorption-controlled kinetic model. Immunoglobulins (or antibodies) are a special class of proteins that are produced by mature B-lymphocytes and plasma cells in the bone marrow of vertebrates in response to external toxins (or antigens) such as, bacteria, viruses, etc. The unique immune response of each immunoglobulin to a particular antigen is used for detecting the presence of toxins in vitro by the well-known process of immunoassay.
In our research, we use nanometer sized colloidal gold particles to tag the antigens after the antigens are selectively captured by the antibodies immobilized on a glass surface. The detection is rapidly made visible to the unaided eye (see Fig. 2) by the reduction of silver ions on the gold nanoparticles in the presence of an initiator. Amplification of the gold signal by 10 to 100 times is readily achieved. The entire detection procedure is a multi-step process (as shown in Fig. 3) and requires optimization with respect to time and concentration in each step. We investigate the mass-transfer characteristics in these immunoassays by studying the diffusion and adsorption kinetics of antibodies/antigens in each step. This study is used to capture the kinetic behavior of the system and describe it using a simple kinetic model which optimizes the overall performance of the immunoassay. These optimized immunoassays are ideal for diagnostic applications.

Figure 2: Schematics of the immunoassay protocol.
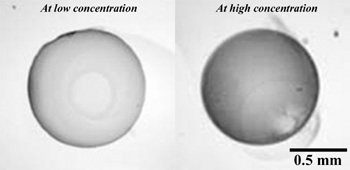
Figure 3: Optical images of silver enhanced spots collected at two different experimental conditions.
Publications
