Yeon Hwang
| yhwang@snut.ac.kr PhD, Materials Engineering |  |
Research Focus: Convective Assembly of Antireflective Silica Coatings
Optical surfaces coated with a film (or multiple films) to reduce reflectance losses provide benefits to a wide variety of technological applications ranging from photovoltaics (solar cells, photodetectors) to display devices such as thermochromic windows, CRTs, eye wear and more. An ideal homogeneous AR coating achieves effectively 100% transmission or 0% reflection at a specific wavelength by optimizing its thickness, l, and refractive index, nc. For brevity, nc must meet the condition of nc =(n0ns)1/2 for zero reflectance. Here, n0 and ns are the refractive indices of the air and the substrate, respectively. If glass substrate ( ns = 1.50) is used, then nc has to be 1.23. Natural or synthetic materials having such low refractive indexes are either rare, expensive to obtain or synthesize, or difficult to manipulate in thin film form. However, porous or microscopically layered heterogeneous (nanoporous) materials can quite easily achieve an effective volume averaged refractive index approaching 1.23. If the pore size is much smaller than the visible wavelengths, the effective n of the nanoporous film is given by an average over the film.
One approach to making porous films is to chemically leach out material from the surface of a substrate to produce small pores; this process was incidentally first employed by Fraunhofer in the 1820s, and today is commonly used for solar cell development. The other technique is to deposit porous films on to the substrate. Sol-gel coating is a commonly used technique, as is sputtering (DC or ion-beam), which is used to deposit various kinds of materials to a wide variety of substrates. Various methods such as plasma-enhanced chemical vapor deposition (PECVD), laser ablation, phase separation and dissolution (of copolymer or polymer blend films), electrostatic deposition have also been used to make antireflective materials.
From the standpoint of simplicity and commercial applicability, rapid and controlled deposition of AR coatings coupled with minimal substrate preparation are desireable. We have elected to take the route of bottom up controlled convective assembly of AR silica nanoparticle coatings. The figures below illustrate the effectiveness that unmodified colloidal silica particles can have on plain glass surfaces. No polyelectrolytes or chemical agents were used, and the silica particles were deposited by evaporative self assembly.
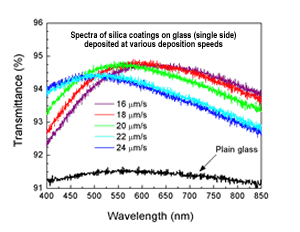
Figure 1: Measured transmittance of a glass microscope slide, and glass slides coated with silica nanocoatings deposited at different deposition speeds. Note that the %T scale is 91-96% to show in more detail the fine control over antireflectance provided by the deposition speed (which controls film thickness). Just ~75 nm particles with no additives provides up to ~70% AR.
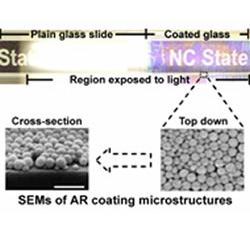
Figure 2: Comparison of uncoated and silica-coated glass microscope slides viewed under strong illumination from above. The SEMs show the representative coating microstructure composed of mixed large and small SiO2 particles (SEM scale bar = 400 nm).
Using just a single type of silica particle, ~70 nm (like shown at above, left), the reflectance per surface could be reduced by up to 70%. In order to further increase the coating porosity and decrease the coating refractive index while maintaining consistent quarter-wave coating thicknesses, binary mixtures of the large and small silica particles were used to break up crystalline close packed structure of the coatings, while maintaining the average film thickness. These mixed particle coatings reduced the reflectance per surface by up to 88%, but were still composed of unmodified silica particles deposited simply and economically by convective assembly. A demonstration of the effectiveness of these AR coatings is shown in the figure on the bottom right.
Publications
